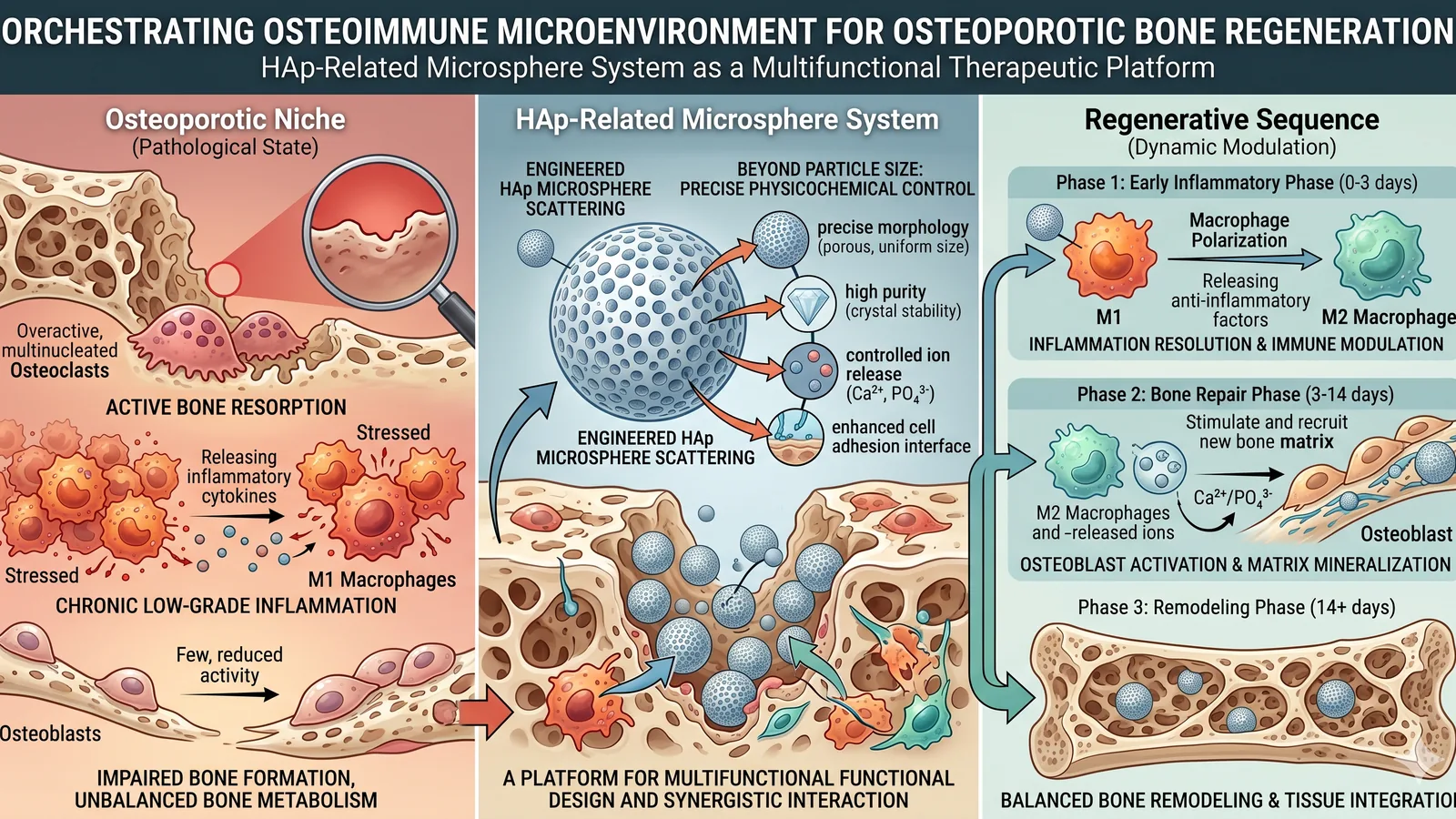
Repairing osteoporotic bone defects involves far more than replacing missing bone volume. The local environment is typically marked by imbalanced bone metabolism, persistent low-grade inflammation, elevated bone resorption, and diminished regenerative capacity. Passive filling and basic osteoconduction alone are increasingly recognized as insufficient; coordinated regulation of the osteoimmune microenvironment, bone resorption–formation balance, and tissue integration has become a central design concern.
Against this backdrop, the way researchers evaluate hydroxyapatite (HAp)-related materials is also evolving. Beyond the well-established role as passive mineral supports, growing attention is directed at how HAp surface chemistry, morphology, ion environment, and composite design may contribute to repair-microenvironment regulation. HAp-related microspheres are of particular interest because they can serve as modular functional units within injectable, composite, or scaffold-based material systems.
Why Osteoporotic Bone Defects Are More Difficult to Repair
Compared with ordinary bone defects, osteoporotic defects are located within a pathological microenvironment that is already out of balance. Recent reviews describe this setting as one involving active bone resorption, impaired osteogenic capacity, persistent inflammatory signaling, and insufficient local support for regeneration. A strategy based only on physical support or one isolated mechanism is therefore unlikely to fully address the complexity of these defects.
From a material-development perspective, this raises the bar for evaluation. A promising bone repair material needs to do more than provide osteoconductive support—it should interact constructively with the host response during the early repair phase, help sustain a favorable local environment, and support the transition from inflammation resolution to new bone formation and remodeling.
HAp Is Being Evaluated Beyond Passive Osteoconduction
HAp has long been regarded as a classic bone repair material, valued for its biocompatibility, similarity to the mineral phase of bone, and osteoconductive behavior. A recent systematic review covering in vivo studies from 2015 to 2025 confirms that HAp remains actively studied in bone remodeling, regeneration, and replacement. What has changed is the depth of inquiry: rather than asking whether HAp can fill and support, researchers now focus on how composition, purity, pore architecture, crystallinity, surface properties, and morphology influence in vivo repair responses.
This deeper inquiry is closely tied to advances in osteoimmunology. Bone healing is now widely understood as a dynamic cascade—immune activation, inflammation resolution, cell recruitment, matrix deposition, mineralization, and remodeling—rather than a single-step gap-filling event. Within this framework, the traditional view of HAp as a passive bone-like mineral is giving way to a broader recognition: its interface with cells, body fluids, and surrounding matrix may actively shape early host responses and downstream tissue integration.
For material suppliers, the implication is clear: evaluating HAp microspheres requires looking past the “is it HAp?” question to examine whether the microspheres deliver stable purity, controlled morphology, reproducible particle-size distribution, and surface characteristics suited to downstream formulation or scaffold integration.
Why Microsphere-Based Systems Matter
Microsphere systems have several advantages in irregular bone defect repair. They can adapt more easily to complex spaces, serve as functional units in composite matrices, and support injectable or printable material strategies. Compared with conventional block-like scaffolds, microspheres often offer greater engineering flexibility for active-component loading, interfacial contact, formulation adjustment, and multi-phase material design.
A distinction should be drawn between HAp microspheres as a material component and engineered multifunctional microsphere systems as a broader platform. Direct evidence for sequential osteoimmune modulation currently comes mainly from composite or engineered microsphere studies, not from single-component HAp microspheres alone. That said, these studies offer valuable design rationale for HAp-related systems: microspheres can serve as carriers, interface modifiers, mineral-phase contributors, or structural building blocks within a coordinated regeneration strategy.
A 2026 study in Journal of Orthopaedic Translation, for example, described multifunctional microspheres designed to intervene sequentially in inflammation, oxidative stress, and bone remodeling balance during osteoporotic bone defect repair. The study was not a direct demonstration of HAp microspheres alone, but it illustrates why microsphere platforms are increasingly discussed in relation to osteoimmune timing and microenvironment reconstruction.
Material Development Implications: More Than Particle Size
Particle size matters, but it is only one part of HAp-related microsphere development. Purity, particle-size distribution, porosity, surface morphology, crystallinity, degradation behavior, and lot-to-lot consistency can all affect interface behavior and downstream repair performance. For upstream material suppliers, these parameters are not just analytical details; they influence the development space available to medical device, scaffold, injectable, and biomanufacturing teams.
A narrower particle-size distribution can improve formulation uniformity and processing consistency. Stable purity control helps reduce uncertainty from unwanted phases or impurities. A carefully designed surface or pore structure may improve contact with cells, body fluids, and extracellular matrix. These features do not automatically translate into superior clinical outcomes, but they form an important material foundation for high-quality downstream research and development.
For 3D-printing bioinks, composite sustained-release carriers, injectable filler matrices, or multi-phase bone repair scaffolds, material consistency often determines how far a formulation can be optimized. HAp-related microspheres should therefore be evaluated as engineered inputs for a broader system, rather than isolated functional units.
From Research Trend to Translational Material Design
The evaluation of bone repair materials has moved beyond demonstrating single-component efficacy. The current benchmark is whether a material system can perform synergistically within a complex microenvironment. Ion release, immune modulation, anti-inflammatory behavior, antioxidant strategy, osteogenic support, matrix mimicry, and structural integration are increasingly integrated within a single design framework, displacing the earlier convention of evaluating materials solely on osteoconductive performance.
For HAp-related microspheres, this points to a practical role as composite-material platforms, functional mineral units, or interface-optimization modules in next-generation bone repair systems. The current evidence base is still largely built on material research, animal studies, and mechanistic exploration; careful standardization and long-term validation remain necessary before broader clinical translation.
Within that boundary, high-quality HAp-related microspheres may provide a stable material basis for bone repair composites and tissue-engineering scaffolds, especially when osteoimmune modulation and microenvironment remodeling are treated as core design priorities.
Conclusion
Repairing osteoporotic bone defects requires structural reconstruction and microenvironment remodeling to proceed in tandem. As osteoimmunology and tissue engineering continue to develop, the value of HAp extends beyond traditional osteoconduction toward a more interface-oriented and system-level material role.
The relevance of HAp-related microspheres will depend on whether they can be designed, characterized, and integrated with sufficient precision. High purity, controllable morphology, stable batch consistency, and compatibility with composite systems are the foundational requirements for their application in advanced bone repair material development.
References
- Fendi F, et al. In vivo studies of hydroxyapatite for bone remodeling, regeneration, and replacement: A bibliometric analysis (2015–2025) and systematic review. F1000Research. 2026;15:543. DOI: 10.12688/f1000research.178985.1.
- Lu X, Zhang A, Zhang C, et al. Targeting the osteoporotic bone microenvironment: Mechanistic insight and therapeutic biomaterials for accelerating bone regeneration. Bioactive Materials. 2026;62:96–122. DOI: 10.1016/j.bioactmat.2026.02.024.
- Luo Z, Ma J, Yang Y, et al. Engineering multifunctional microspheres for sequential regulation of osteoimmune microenvironment and bone remodeling balance to promote regeneration of osteoporotic bone defects. Journal of Orthopaedic Translation. 2026;57:101054. DOI: 10.1016/j.jot.2026.101054.